 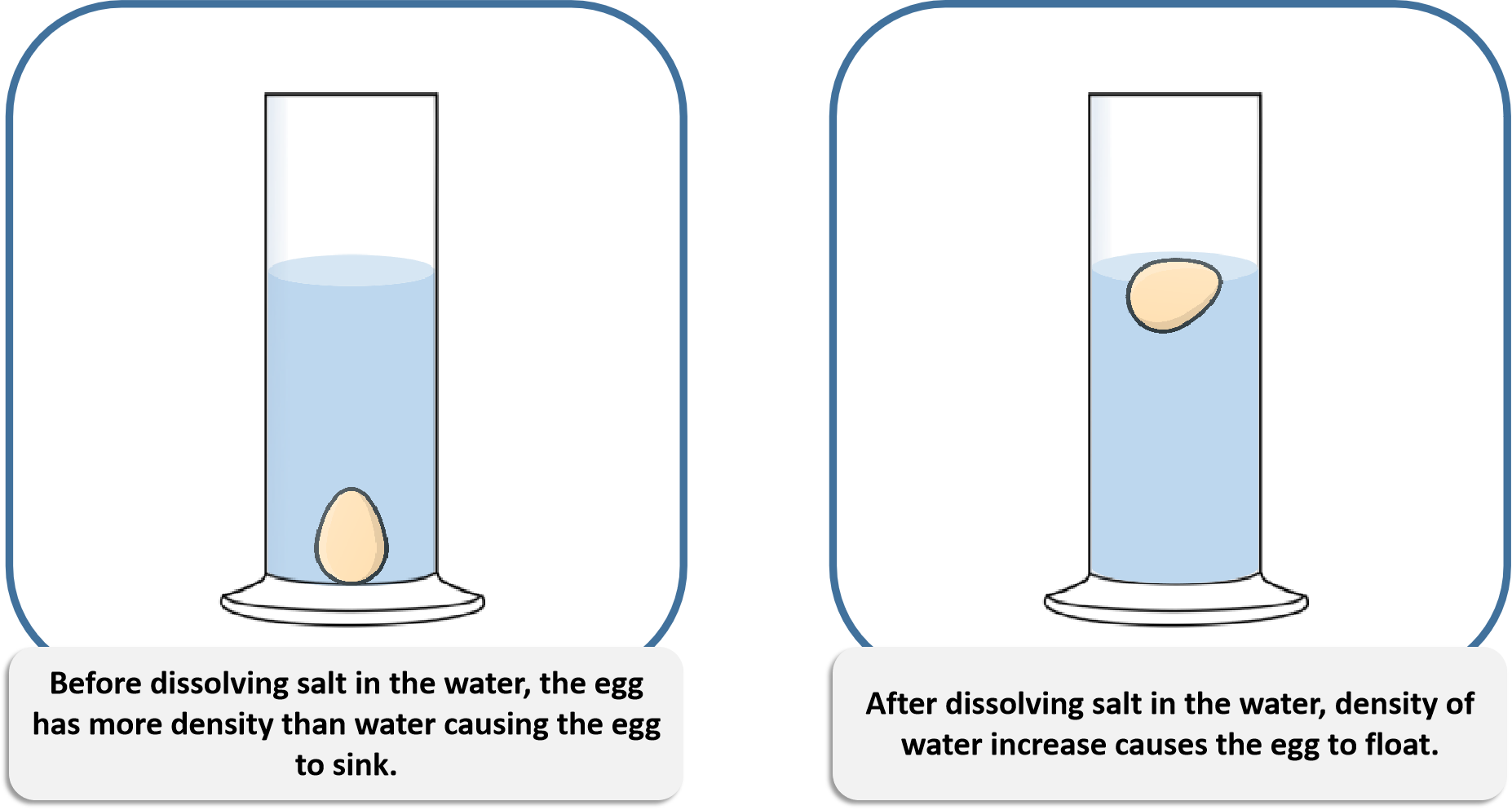
Therefore, it is difficult for steel ships to float if they are made only of steel. The density of water, however, is much lower, at 1g/cm 3. Its overall density is approximately 7.85g/cm 3. As less dense things float, the balloon will rise.  
However, as soon as the particles are heated, they move more vigorously, and as they bump into each other more often, they become less dense. Did you know that a hot-air balloon has to be heated before it flies? This is because the particles inside the balloon are just as dense as the particles outside the balloon at the beginning. How exactly? Look at the diagram on the right. They work the same way: using the differences in density to hurl the balloon up. A hot air balloon is just like those you ride in amusement parks. Once upon a time, when aeroplanes were yet to be invented, the only way to soar through the skies was to ride in a hot-air balloon. Let's get to a topic with no sums: how vehicles work.īalloons The engine of a hot-air balloon You can refer to the diagram on the left, which shows a container with all the solids and liquids in it.īalloons, ships and submarines Since Solids W and Y are both on the bottom, they are on the same level-they both sink in liquid A. Solid X < Liquid B < Water < Solid Z < Liquid A < Solid W < Solid Y We will round off to three significant figures here. Note that we use division rather than fractions here. If all these things are put into a measuring cylinder (or any container for that matter), and nothing dissolves, what will be the order of their altitude, from top to bottom? The following is a table of the name, mass and volume of some liquids and solids. Unfortunately, we need a bit of math here. In other words, whether an object will float or sink in a certain liquid (or gas) depends on their density with relation to the density of the liquid. On the contrary, the feathers' density is probably less than 1g/cm 3, which makes them float. The density of water is 1 g/cm 3, so if the density of the lead is more than 1g/cm 3, it has to sink. As there is the same mass stuffed in a smaller space, it must be denser. While their mass may be the same (1kg), the volume of lead is a lot smaller than that of feathers. But what if you put the lead and feathers into a pond? Will they both sink?Īctually, no. What does all that have to do with floating or sinking? Here's an old riddle: which one is heavier, 1kg of lead or 1kg of feathers? Yes, that's right, they are the same. Note: You can also swap them around like you do in maths to find the mass or volume when you have the density.įloating and sinking This formula is very important, so we will put it here:ĭensity = Mass Volume By now you may have deduced that the method of finding density is mass over volume. What are the units of density then? The units of density are always constructed by the unit of the mass, a forward-slash, and then the unit of the volume. That is also the density in there: density is the amount of matter in a specific space the volume is usually represented by an invisible '1'. Remember that in your room, there are (on average) 4 kg in every cubic meter. Then, in every average cubic metre, there would be 30 kg ÷ 7.5 m 3 = 4 kg. If your room is 1.5m × 2m × 2.5m, and your mass is 30kg, then there are 30kg in every 1.5m × 2m × 2.5m = 7.5 m 3. Therefore, you are the only thing with mass in it. You are the only one inside the empty, airless room. Imagine you are in an enclosed room, say your bedroom. Mass is measured in the units kilogram, gram and milligram. Do you remember that there are always spaces between particles? 'Mass' does not include the spaces between the particles, while 'volume' does. 'Mass' means how much of matter there is. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
